- AI
- Molecular Imaging
- CT
- X-Ray
- Ultrasound
- MRI
- Facility Management
- Mammography
FDA Clears New 7T MRI System from Siemens Healthineers
Offering improved image clarity capable of capturing details of subtle pathology, the Magnetom Terra.X 7T MRI system reportedly features the first eight-channel parallel transmit architecture for clinical use.
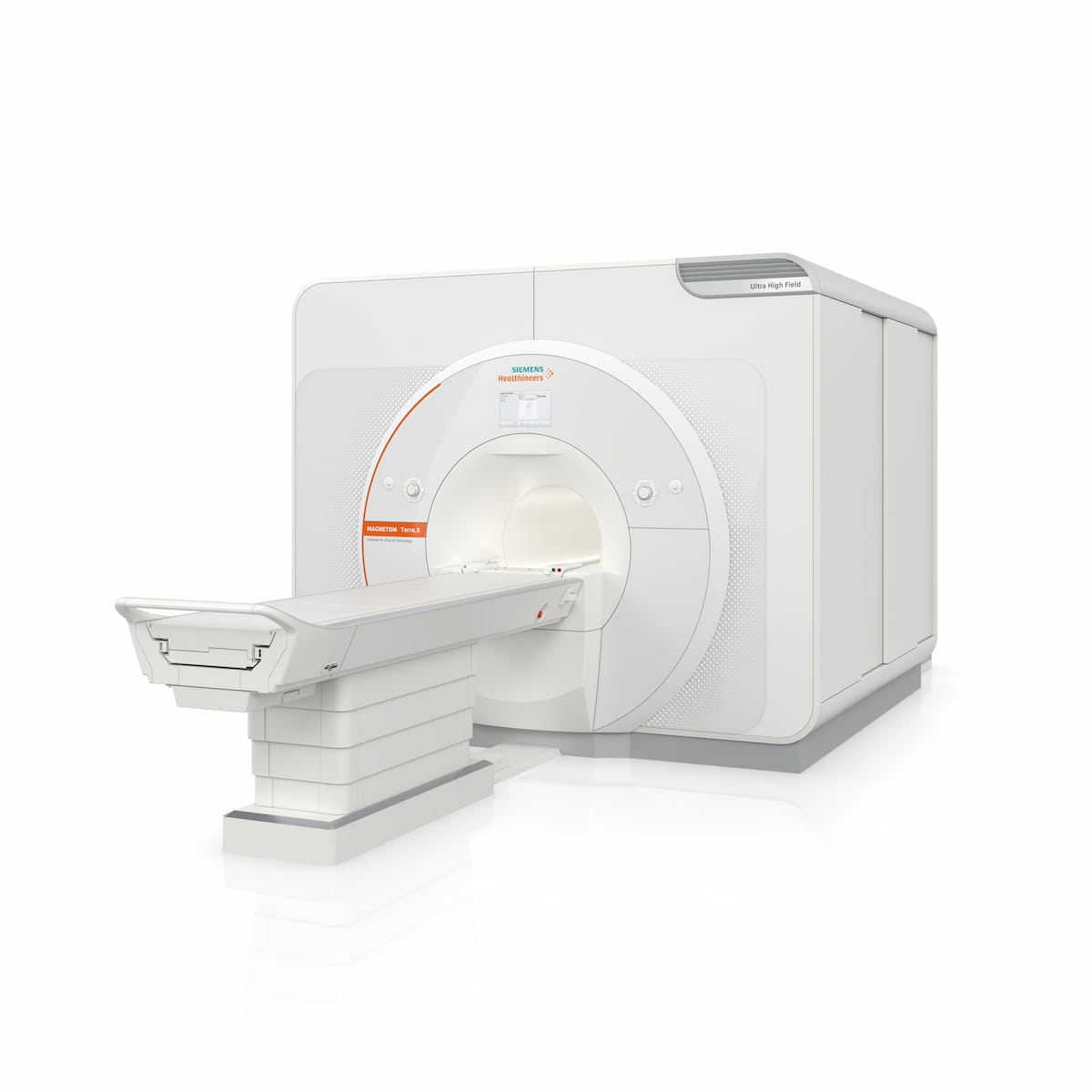
The Magnetom Terra.X., a new 7T magnetic resonance imaging (MRI) system, has garnered 510(k) clearance from the Food and Drug Administration (FDA).
A second-generation successor to the Magnetom Terra, the Magnetom Terra.X system has a number of attributes to provide enhanced 7T imaging, according to Siemens Healthineers, the manufacturer of Magnetom Terra.X.
The Magnetom Terra.X system, recently cleared by the FDA, has a number of features including the first eight-channel parallel transmit architecture for clinical use, according to Siemens Healthineers, the manufacturer of the 7T MRI system. (Image courtesy of Siemens Healthineers.)
The company said key features of the device include:
• the first eight-channel parallel transmit architecture for clinical use;
• deep learning image reconstruction specifically geared to 7T;
• improved diffusion imaging via a high-performance gradient system; and
• accelerated image acquisition capabilities that can facilitate high-resolution brain and knee exams in less than 20 minutes.
“The MAGNETOM Terra.X builds on our engineering achievements as pioneers in 7T MRI by providing the ultra-high-field community with the necessary tools to offer higher-quality patient care not just in neurological imaging, but also in the knee,” said Katie Grant, vice president of magnetic resonance at Siemens Healthineers North America.
Siemens Healthineers added that the FDA clearance includes upgrades of installed Magnetom Terra systems to the Magnetom Terra.X.
European Society of Breast Imaging Issues Updated Breast Cancer Screening Recommendations
April 24th 2024One of the recommendations from the European Society of Breast Imaging (EUSOBI) is annual breast MRI exams starting at 25 years of age for women deemed to be at high risk for breast cancer.
New AI-Powered Ultrasound Devices May Enhance Efficiency in Women's Imaging
April 19th 2024One of the features on the new Voluson Signature 20 and 18 ultrasound devices reportedly uses automated AI tools to facilitate a 40 percent reduction in the time it takes to perform second trimester exams.